Nuvaxovid
The subunit that is used here as vaccine is the spike protein S of SARS-CoV-2. The Technical Advisory Group for Emergency Use Listing listed Nuvaxovid NVX.

Takeda Gains Approval In Japan For Nuvaxovid Covid 19 Vaccine For Prim
Find detailed technical information such as the product monograph and.
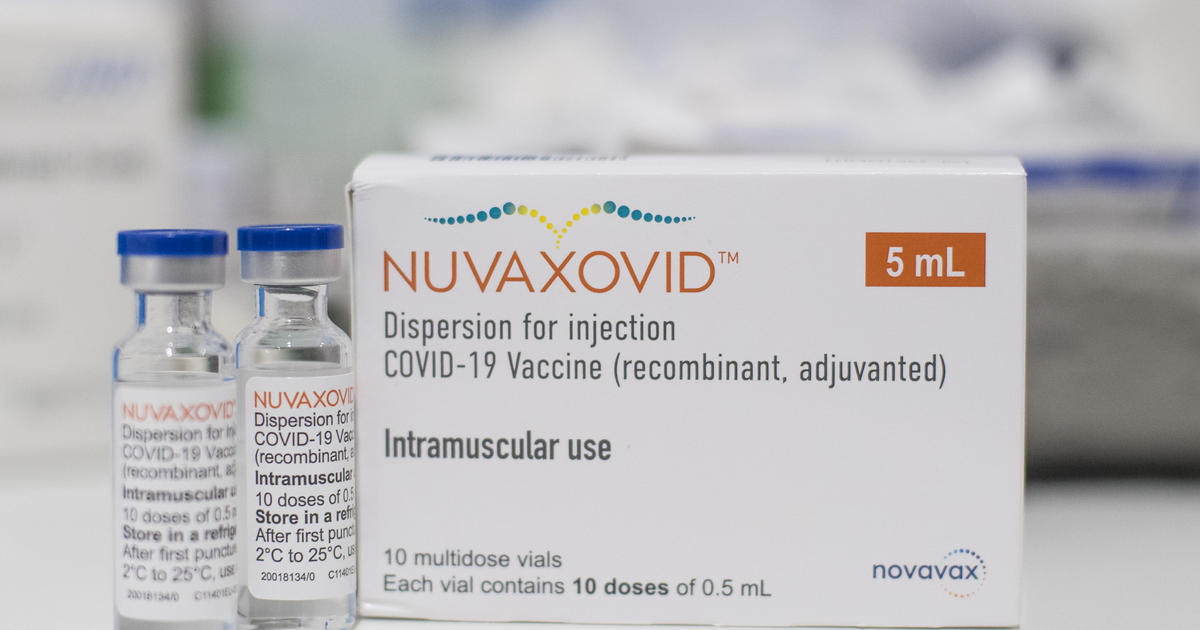
. COVID-19 Vaccine recombinant adjuvanted 2. It is recommended that. Name of the medicinal product.
This is a multidose vial. Information about the COVID-19 vaccine Nuvaxovid approved by the MHRA on 03 February 2022. This webpage was updated on 28 September 2022 to ensure consistency of formatting.
It is recommended to administer the second dose 3 weeks after the first dose see section 51. Around 7000 doses of Nuvaxovid have already been administered in Sweden. Novavax Nuvaxovid COVID-19 Vaccine Description 2022.
The booster dose is given 3 months or more after the primary course. Nuvaxovid is composed of purified full-length SARS-CoV-2 recombinant spike S protein that is stabilised in its prefusion conformation. Publicerad idag 0702.
Active immunisation to prevent coronavirus disease 2019 COVID. This protein mediates the binding of the virus to the cell surface and is thus responsible for the infection of. Nuvaxovid will be given to you as two separate 05 mL injections.
Nuvaxovid is given as two injections usually into the muscle of the upper arm 3 weeks apart. Nuvaxovid is given as two injections usually into the muscle of the upper arm 3 weeks apart. Medical News Today has contacted over 20 experts for comment on the potential side effects of the Novavax COVID-19 vaccine Nuvaxovid.
Nuvaxovid dispersion for injection. Nuvaxovid contains a version of a protein found on the. The agency said that younger people who had recently been vaccinated with Nuvaxovid had no.
Det eftersom att data från. Nuvaxovid SARS-CoV-2 rS with matrix M adjuvant NVX-CoV2373 was approved for the following therapeutic use. Your doctor pharmacist or nurse will inject the vaccine into a muscle usually in your upper arm.
Esimerkiksi aiemmin sairastettu koronavirustauti ei estä rokotuksen antamista. A booster dose of Nuvaxovid may be given to people aged 18 years and. Qualitative and quantitative composition.
Novavax COVID-19 vaccine Nuvaxovid CovoVax NVX-CoV2373 is a protein-based vaccine engineered from. The European Commission will now fast. Rokotteesta ei myöskään ole haittaa vaikka.
The World Health Organization issued an emergency use listing EUL for Nuvaxovid TM following its assessment and approval by the European Medicines Agency EMA earlier. Det proteinbaserade covid-19-vaccinet Nuvaxovid inte ska ges till personer som är 30 och yngre meddelar Folkhälsomyndigheten. The Nuvaxovid NVX-CoV2373 Novavax vaccine is a recombinant spike S protein nanoparticle vaccine combined with the Matrix-M adjuvant.
The Novavax Nuvaxovid COVID-19 vaccine was authorized for use in Canada under the Food and Drug Regulations. As of the time of publication they. Sverige Covid-19-vaccinet Nuvaxovid skulle erbjudas till personer som var tveksamma till vaccinationen.
Novavax can also be used as a booster dose in people aged 18 years and older. Nuvaxovid is administered intramuscularly as a course of 2 doses of 05 mL each. Web Nuvaxovid Novavax is approved and available for use as a primary course in people aged 12 years and over.
Nuvaxovid is a vaccine for preventing coronavirus disease 2019 COVID-19 in people aged 12 years and older. Beslutet är temporärt och gäller från. Some people are also.
Folkhälsomyndigheten rekommenderar att det proteinbaserade covid-19-vaccinet Nuvaxovid inte ges till personer som är 30 år och yngre. Nu stoppar Folkhälsomyndigheten användningen bland personer som är 30. COVID-19 Vaccine recombinant adjuvanted 2.
The Summary of Product Characteristics is a description of a medicinal. Nuvaxovid-rokote sopii lähes kaikille aikuisille. On December 20 2021 the.

Ema Recommends Nuvaxovid For Authorisation In The Eu Certifico Srl
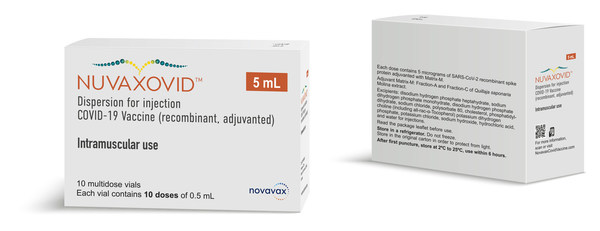
Novavax Announces Shipments Of Its Covid 19 Vaccine To European Union Member States Feb 23 2022

Novavax Announces Shipments Of Its Covid 19 Vaccine To European Union Member States Feb 23 2022

Novavax Nuvaxovid Covid 19 Vaccine Granted Expanded Conditional Marketing Authorization In The European Union For Use As A Booster For Adults Aged 18 And Older Sep 12 2022
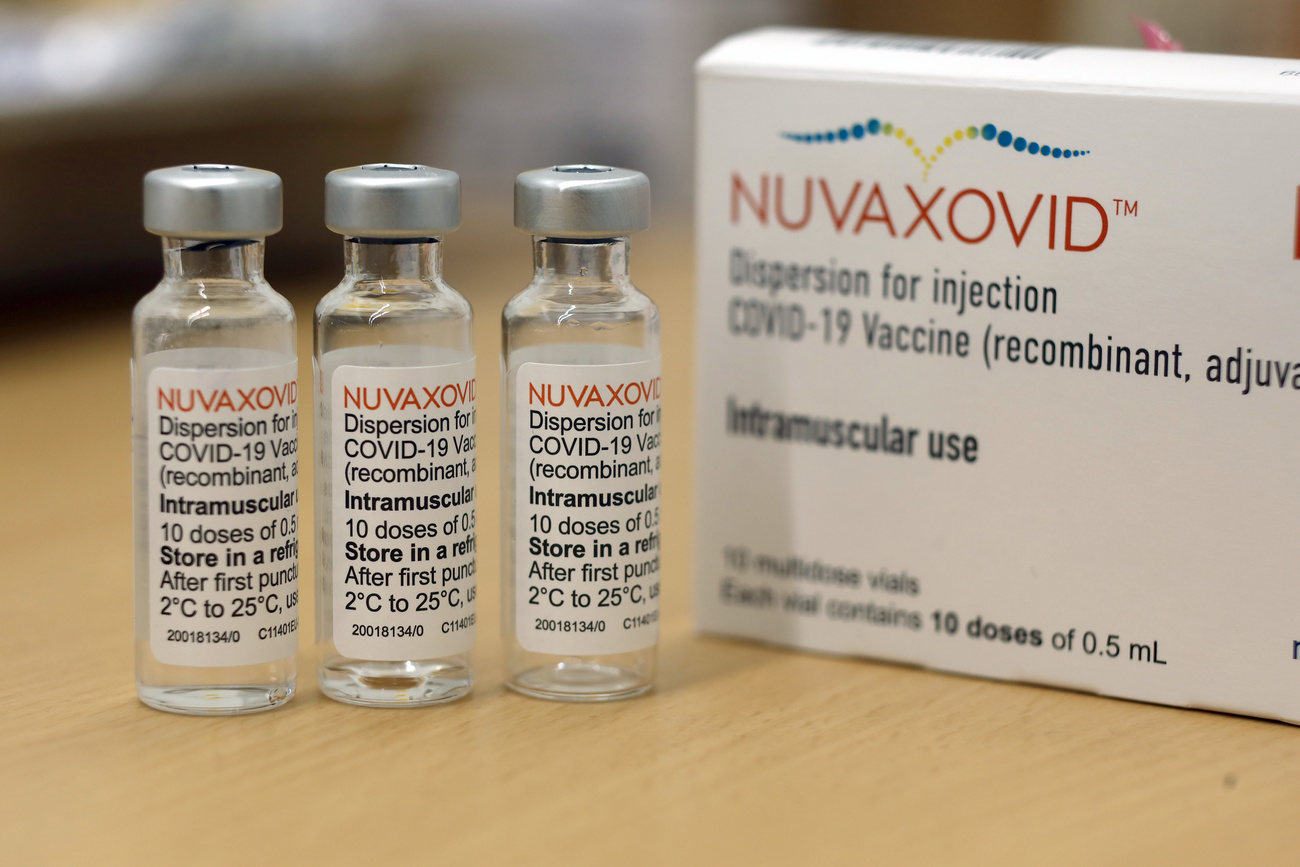
Switzerland Approves Its First Protein Based Covid Vaccine Swi Swissinfo Ch

Novavax Covid 19 Vaccine Nuvaxovid Data On Side Effects

Webcast An Overview Of The Novavax Nuvaxovid And Medicago Covifenz Covid 19 Vaccines Youtube

Novavax Requests Expanded Emergency Use Listing With Who For Nuvaxovid Covid 19 Vaccine For Adolescents Aged 12 Through 17 Eatg

What To Know About The New Novavax Vaccine For Covid 19 The Hill

Novavax Eyeing The Covid Vaccine Hesitant And Kids Unveils New Education Campaigns As Nuvaxovid Nears Us Finish Line Fierce Pharma
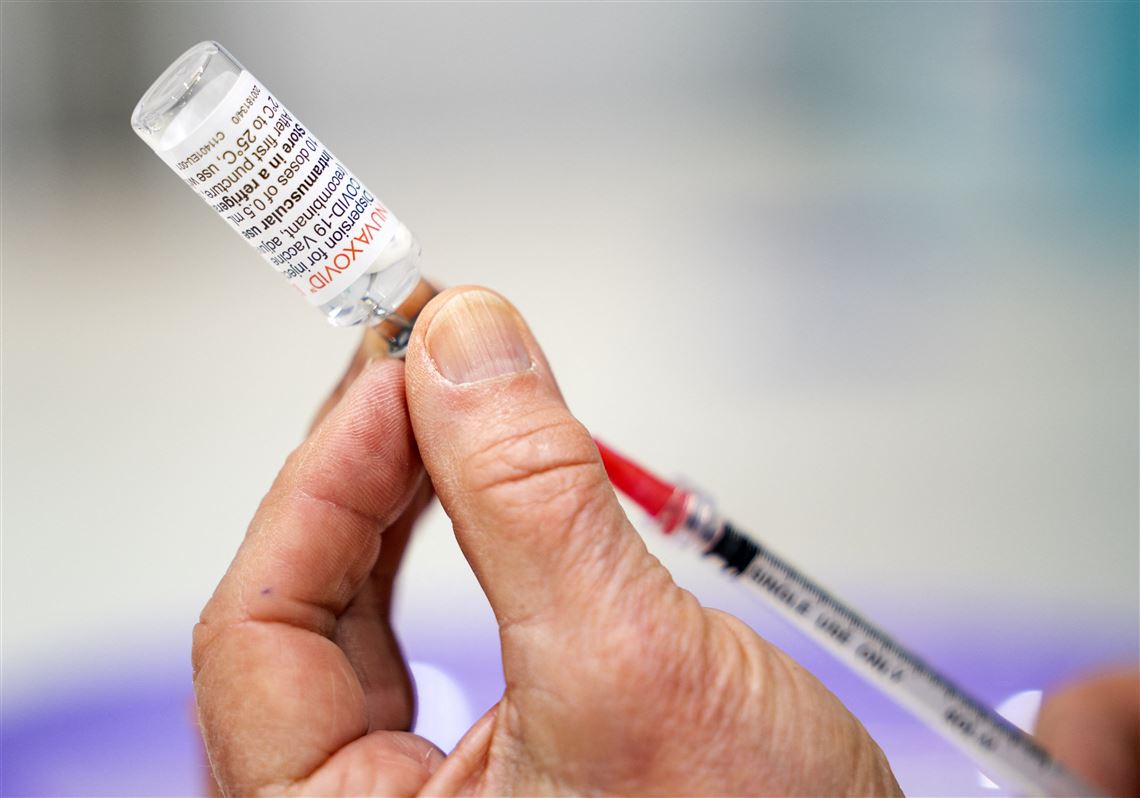
Allegheny County Health Department Will Start Administering Novavax Vaccine Pittsburgh Post Gazette

Novavax Stock Looks Like A Good Value With Its New Combined Vaccine

News Nuvaxovid Novavax Protein Based Covid 19 Vaccine Available On A Limited Basis In Germany Paul Ehrlich Institut

Novavax S Covid 19 Vaccine Approved For Canadians 18 And Older Cbc News
Nuvaxovid The New Subunit Sars Cov 2 Vaccine Mci Innsbruck

Canadian Trademarks Details Nuvaxovid 2163962 Canadian Trademarks Database Intellectual Property And Copyright Canadian Intellectual Property Office Innovation Science And Economic Development Canada

Novavax Says Initial 1m Doses Of Nuvaxovid Covid 19 Vaccine Are Available In Uk

